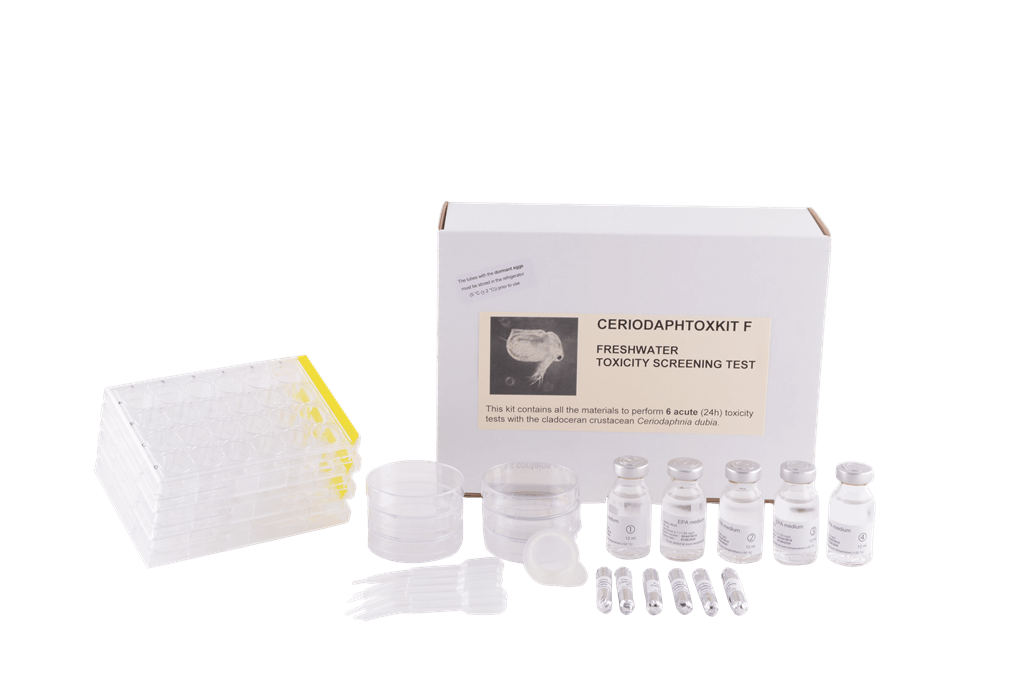
Ceriodaphtoxkit F
The acute Ceriodaphnia dubia toxicity test is intended for toxicity screening of chemicals, effluents, surface waters, wastewaters, groundwaters, sediment pore waters and elutriates
In our Ceriodaphtoxkit F, you find all the necessary materials to perform six acute 24h mortality tests with the freshwater crustacean Ceriodaphnia dubia. The Daphnia mortality test is cost-effective, culture-independent, user-friendly and highly standardised, it adheres to International Standard USEPA 2002.0
How to order?
FRESHWATER CERIODAPHNIA TOXICITY TEST
Test criterion
The acute toxicity test with Ceriodaphnia dubia is a 24h bioassay based on the mortality of the test organisms, with calculation of the 24h EC50.
Contents of the Ceriodaphtoxkit F
- Tubes with ephippia (dormant eggs), which you can hatch on demand to perform the freshwater Ceriodaphnia toxicity test.
- Concentrated hatching and toxicant dilution medium, hatching and test containers and micropipettes for the transfer of the organisms.
- Short Bench Protocol and standard operating procedure brochure with easy-to-follow instructions and detailed illustrations.
- Data scoring sheets and a Quality Control specification sheet with batch number of the ephippia and the media.
- Tubes with ephippia (dormant eggs), which you can hatch on demand to perform the freshwater Ceriodaphnia toxicity test.
- Concentrated hatching and toxicant dilution medium, hatching and test containers and micropipettes for the transfer of the organisms.
- Short Bench Protocol and standard operating procedure brochure with easy-to-follow instructions and detailed illustrations.
- Data scoring sheets and a Quality Control specification sheet with batch number of the ephippia and the media.
All test materials and equipment included in the Ceriodaphtoxkit F are available separately.
Benefits of the Ceriodaphnia toxicity test
- We produce high-quality ephippia in strictly controlled conditions. This way, we preclude variability associated with recruitment/maintenance of live stocks in conventional bioassays.
- The quality of the standardised test medium is highly uniform, thanks to simple dilution of concentrated solutions of selected salts with deionised water.
- A Quality Control test with a reference chemical is described in detail, so you can easily check accuracy and reproducibility.
- You can hatch the ephippia on demand, which eliminates the need and the costs of continuous culturing and maintenance of your test organisms.
- The Ceriodaphnia toxicity test requires minimal space and equipment:
- dissection microscope
- incubator with lateral lights
- conventional laboratory glassware
- If you store the ephippia properly, you can prolong their shelf life for several months and reduce test scheduling constraints.
- Simple handlings and scorings.
- Total performance time of the Daphnia toxicity assay is approximately 1 hour.
- We can provide you with a computer program for easy Toxkit data treatment.
The sensitivity of the daphnids hatched from ephippia is comparable to the sensitivity of Ceriodaphnia originating from laboratory cultures.
- The Ceriodaphtoxkit F is used in many environmental laboratories and research institutes worldwide.
- You can find a substantial number of publications, posters, reports and reviews for various uses.
- Our Ceriodaphnia toxicity test adheres to International Standards USEPA 2002.0. for regulatory testing with Ceriodaphnia dubia.
Interested in buying Ceriodaphtoxkit F?
It contains all the materials you need to perform six 24h mortality tests with the freshwater crustacean Ceriodaphnia dubia.